pH & Ocean Acidification
Principles of pH
ASSOCIATED READING: pg 51-53 in Campbell Biology (Figure 3.11 especially). pg 52-54 in OpenStax.
**Things you should be able to do:
- Memorize that an acid is an H+ "donor". It releases H+ into a solution of water.
- Write numbers in scientific notation. Focus on the fractions (e.g. 1/100).
- Describe how the concentration of hydronium (or H+) relates to the "pH" of a solution.
- Memorize that "[H+]" is short-hand for "hydrogen ion concentration".
- Given a pH value, predict the [H+].
- Describe how a base ("hydrogen acceptor") affect [H+].
- Understand the degree by which a pH change of 1 corresponds to the abundance of H+ ions in a solution of water. NOTE: a change in hydronium is equivalent to a change in H+.
How ocean animals make their shells
Click here to watch a very short video describing the process of calcification in ocean-dwelling organisms. **Things you should be able to answer from the video:
- What two substances are commonly used by ocean organisms for shell building?
Biological Consequences of Ocean Acidification
**Things you should be able to answer from the article linked directly above:
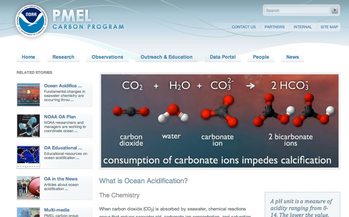
THE CHEMISTRY:
THE CHEMISTRY:
- What minerals are normally very abundant in ocean water and used by organisms to build shells?
- How does acidification affect the availability of these minerals?
- Since the Industrial Revolution, by what percentage has the acidity increased globally in our oceans?
- What are the impacts of ocean acidification on:
- ocean ecosystems
- coastal industries.
- In what way will be water arriving on the West coast be affected during the next 30-50 years.